|
8 Bavarian Health and Food Safety Authority (LGL), Oberschleißheim, Germany.7 Laboratory for Functional Genome Analysis, Gene Center, LMU München, Munich, Germany.6 Gene Center and Department of Biochemistry, LMU München, Munich, Germany.5 Pediatric Rheumatology / Special Immunology, Pediatrics Department, University Hospital Würzburg, Würzburg, Germany.4 Institute of Virology, Helmholtz Center Munich, TUM, School of Medicine, Munich, Germany.3 Institute of Molecular Immunology and Experimental Oncology, University Hospital rechts der Isar, Technical University of Munich (TUM), School of Medicine, Munich, Germany.2 German Centre for Infection Research (DZIF), Partner Site, Munich, Germany. 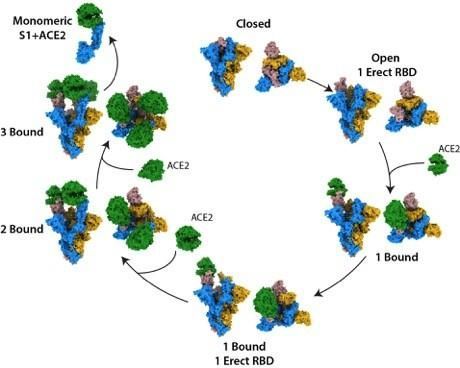
1 Max von Pettenkofer Institute and Gene Center, Virology, National Reference Center for Retroviruses, Faculty of Medicine, LMU München, Munich, Germany.Sources: National Center for Biotechnology Information Nature Florian Krammer, Icahn School of Medicine at Mount Sinai. May 7 Pfizer and BioNTech apply for full approval from the U.S. 26 The companies announce a study to develop a B.1.351-specific booster.Īpril 16 Pfizer says their vaccine may require a third dose within a year of the initial inoculation, followed by annual vaccinations.Īpril 20 Some vaccinated people are professing loyalty to the brand they happened to have received.Īpril 25 Nearly 8 percent of Americans who got initial Pfizer or Moderna shots have missed their second doses.Īpril 28 Pfizer has delivered more than 150 million doses of the vaccine to the United States, and expects to double that number by mid-July. 15 Pfizer and BioNTech register a trial specifically for pregnant women.įeb. However, researchers find that antibodies produced by Comirnaty are somewhat less effective against another variant called B.1.351, first identified in South Africa.įeb. Tests on a variant called P.1, first identified in Brazil, show that Comirnaty will likely work against it as well. January, 2021 Scientists grow concerned about the emergence of fast-spreading variants that might be able to evade antibodies. Each vaccinated person will require two doses. 31 Pfizer expects to produce up to 50 million doses by the end of the year, and up to 1.3 billion doses in 2021. 21The European Union authorizes the vaccine.ĭec. 14 Vaccination begins in the United States.ĭec. Mexico also approves the vaccine for emergency use.ĭec. grants Pfizer-BioNTech vaccine the first emergency use authorization for a coronavirus vaccine in the United States. 10 Saudi Arabia approves the vaccine.ĭec. 9 Canada authorizes the Pfizer-BioNTech vaccine.ĭec. 8 William Shakespeare, age 81, is among the first people to receive a shot of the vaccine in Britain, on the first day of vaccinations for at-risk health care workers and people over 80.ĭec. 2 Britain gives emergency authorization to Pfizer and BioNTech’s vaccine, becoming the first Western country to give such an approval to a coronavirus vaccine.ĭec. 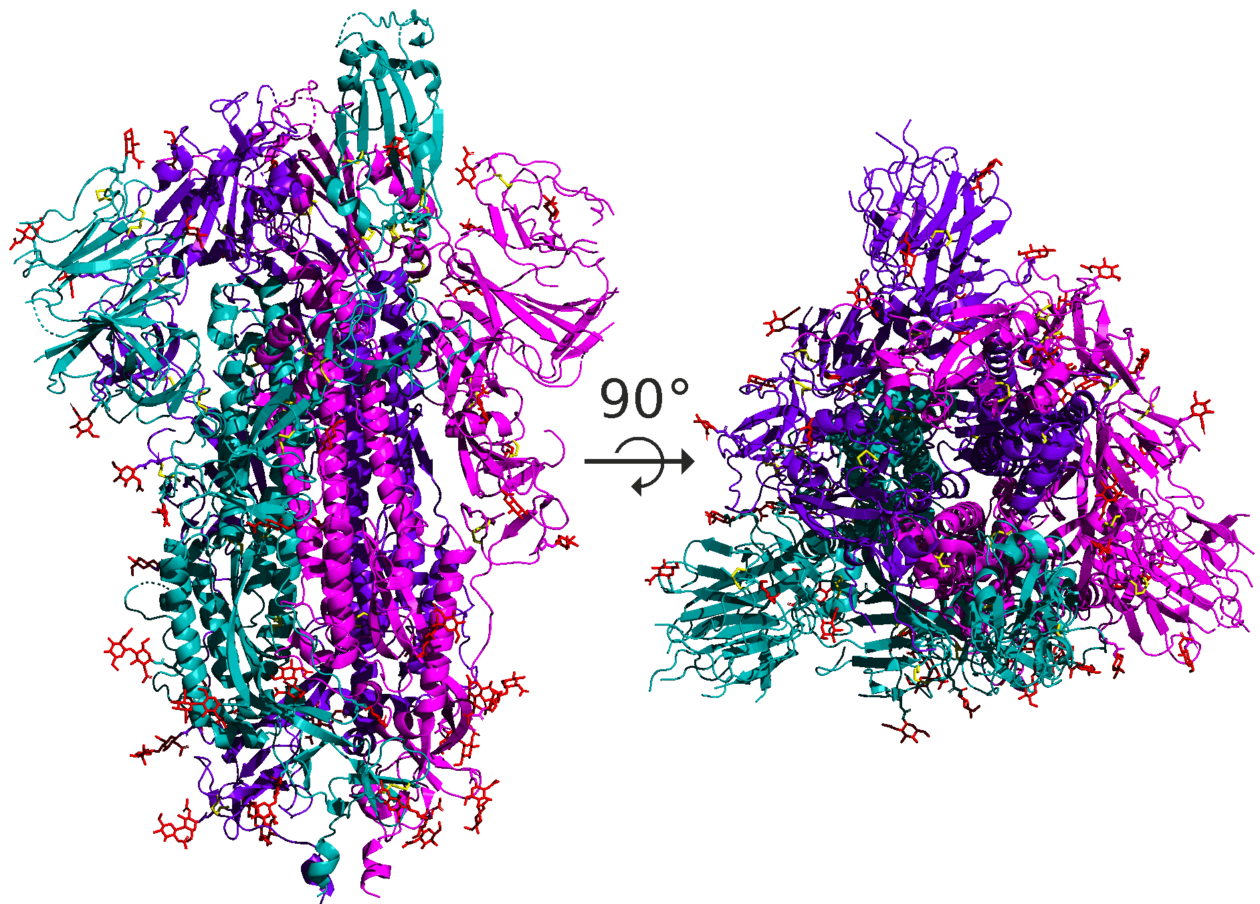  
20 Pfizer requests an emergency use authorization from the F.D.A.ĭec. The final data from the trial shows the efficacy rate is 95 percent. 9 Preliminary data indicates the Pfizer vaccine is over 90 percent effective, with no serious side effects.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
